Flu shots vs. nasal spray vaccines — What’s the difference?
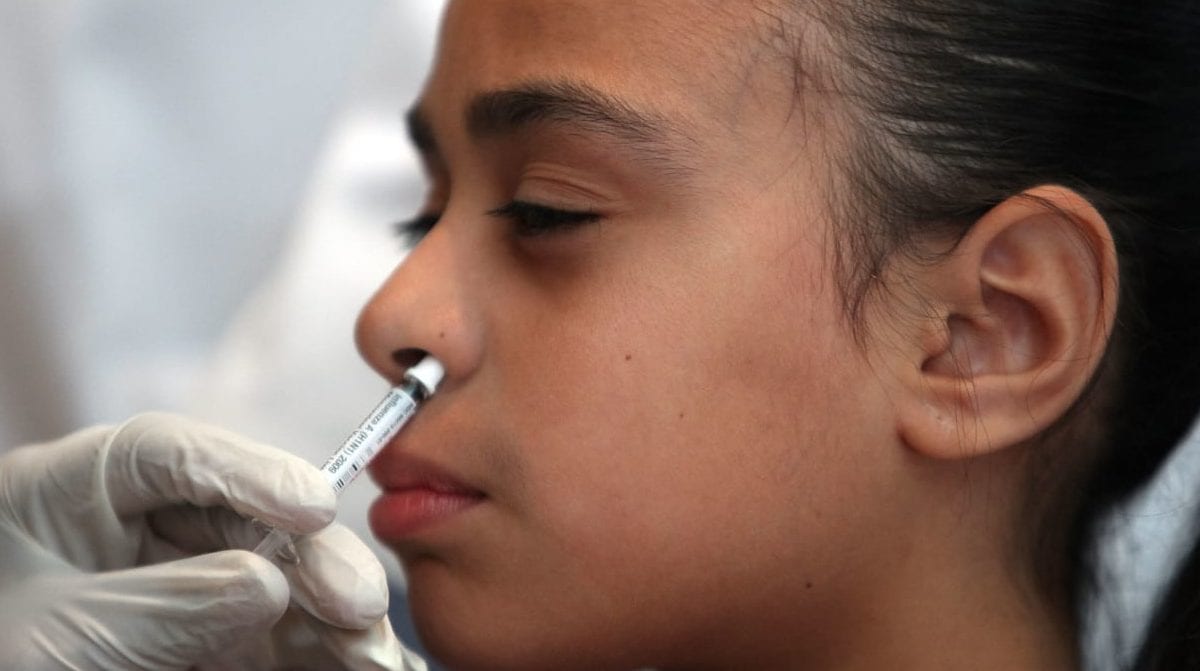
Good news for shot-averse kids and others who cringe at the sight of needles: After a two-year hiatus from the market, the nasal spray flu vaccine has been re-formulated and is back as we enter the 2018-2019 flu season.
FluMist was first licensed in 2003, and was welcomed as a needle-free alternative to the flu shot, which made it especially easy to administer in school clinics. But in 2016, a Centers for Disease Control and Prevention advisory committee stopped recommending FluMist because it didn’t perform well against a particular flu strain, the H1N1 strain. In some cases, the nasal spray vaccine offered only 3-percent protection for children between ages 2 and 17, compared with up to 63-percent protection from the shot.
While we call it “the flu” in general, influenza can actually be caused by various strains, which evolve. Every flu season, there are predominant strains of the virus, so medical experts predict which strains will be most active, and they formulate the vaccines in response. This year, a re-formulated nasal spray is once again an option, and the CDC states on its fact sheet that “no preference is expressed for any influenza vaccine over another”
“The main advantage of the nasal spray is that it’s painless,” says Dr. Robert Segal, co-founder of LabFinder.com, which allows patients to book appointments online. “It’s the best alternative for those who don’t like needles.”
In fact, offering the painless alternative to the shot could help health officials achieve their goal of getting 70 percent of Americans vaccinated. (At last count, only about half of the country gets a flu vaccination, which are recommended for those over six months of age).
Here’s what else you need to know about the return of nasal spray vaccine.
Who Is Eligible For The Nasal Spray?
FluMist Quadrivalent, the nasal spray, is approved for use in non-pregnant individuals who are ages 2 to 49. However, some people with medical conditions should not receive the nasal spray flu vaccine.
Some of those conditions include: Children ages 2 to 17 who are taking aspirin or salicylate-containing medication; people who have weakened immune systems; those who are age 5 and up who have asthma; and others with conditions such as lung disease, heart disease, kidney disease, or neurological or neuromuscular disorders. You can find more information about who is eligible for the nasal spray at CDC.gov.
So, if you’re in the right age bracket without any limiting conditions, how do physicians decide which version to offer?
“Some physicians might prefer the injectable vaccine — in fact, the American Academy of Pediatrics recommends it — but in general, patient preference and vaccine availability drive the process,” explains Dr. Amesh A. Adalja, a board certified infectious disease physician and a senior scholar at the Johns Hopkins Center for Health Security.
As stated above, the American Academy of Pediatrics, in a statement, recommended the shot, saying the academy believes it’s the most effective and that it remains to be proven whether the nasal spray will be effective against H1N1 this year.
An advantage to the nasal spray, though, is that it’s a live vaccine and administered in the nasal passages, Adalja says. Because of that, it may mimic a natural flu infection more closely, which, in turn, could provide better influenza protection, he explains.
Dr. Frank Illuzzi, chief medical officer at CityMD, and a board-certified physician in emergency medicine, recommends patients speak with their medical providers about the different flu vaccine options to determine which one is the best fit for them.
“I encourage people to not delay, and to get a flu vaccine,” Illuzzi says. “The CDC recommends all individuals over the age of six months get a yearly flu vaccine and that is the best protection against the virus.”
For example, during 2016-2017, flu vaccinations prevented an estimated 5.3 million influenza illnesses, 2.6 million flu-associated medical visits and 85,000 flu-associated hospitalizations, according to the CDC.
Is The Nasal Spray As Effective As The Shot?
The short answer: Yes, it’s expected to be, according to the CDC, which isn’t placing preference on any single vaccine.
Flu season runs from October until May, peaking in January and February. Right now, it’s early in the flu season and health professionals have just started tracking flu cases and the types of strains that are most common, Illuzzi explains.
“The CDC and local departments of health keep track of this information and report it back to clinicians on a weekly basis,” he says. “The preliminary results seem promising that this year‘s flu vaccines appear to be a good match.”
The monitoring is done by the CDC’S U.S. Flu Vaccine Effectiveness Network, and similar studies are conducted in the United Kingdom, Canada and Finland, explains Dr. Chris Ambrose, franchise head with U.S. Medical Affairs, respiratory division at AstraZeneca, the maker of the FluMist Quadrivalent. The CDC provides vaccine estimates several times throughout the season, and typically early estimates are reported in February and the full season estimates come in June, Ambrose says.
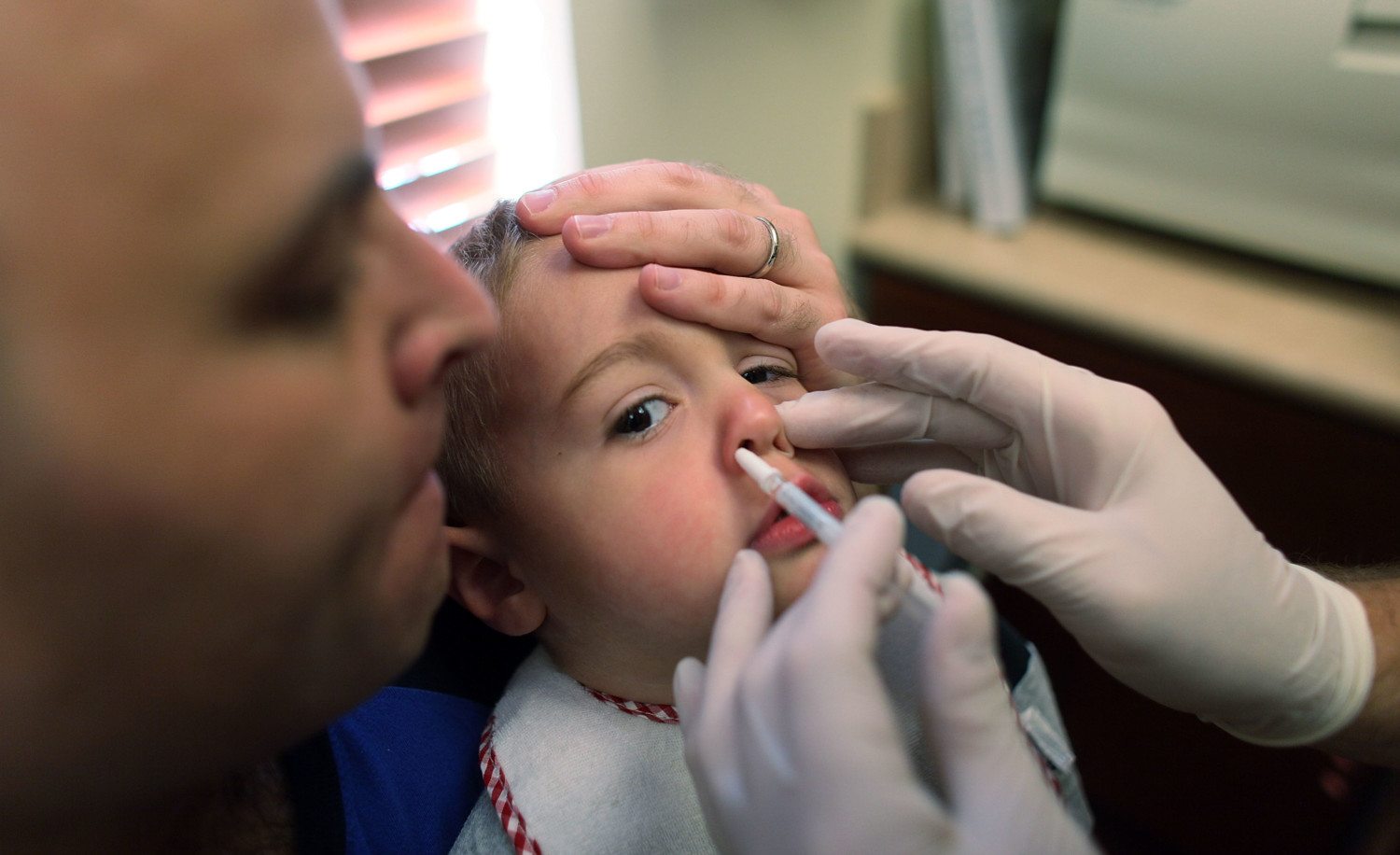
How Does FluMist Work?
The nasal spray vaccine starts working where the flu typically starts, which is in the nose, explains Ambrose. To provide flu protection, it works in your nose, cells and bloodstream, he says. Similar to other live vaccines (think: the chicken pox vaccine), FluMist Quadrivalent contains a weakened live virus. It will not cause the flu, but it does trigger your immune system to build antibodies throughout the body to help fight against the flu virus, Ambrose explains.
Need to get your flu vaccine? Here’s how to find a clinic near you.
Also, if you want to go for the bonus round in your flu-shielding efforts this year, in addition to getting a flu shot, we’ve rounded up some tips on how doctors and nurses stay healthy during flu season.