Does Tapping A Soda Can Really Keep It From Exploding?

For many of us, the habit of tapping the top of a soda can before opening it may be deeply engrained. Tapping it, the thinking goes, will help prevent the fizzy liquid inside from bubbling over the top when you open it.
One British researcher wasn’t content with a “may or may not” situation, and so he decided to put tapping to the test. Carbonated liquids fizz over the top when a can is opened because the escaping carbon dioxide bubbles bring liquid to the top with them.
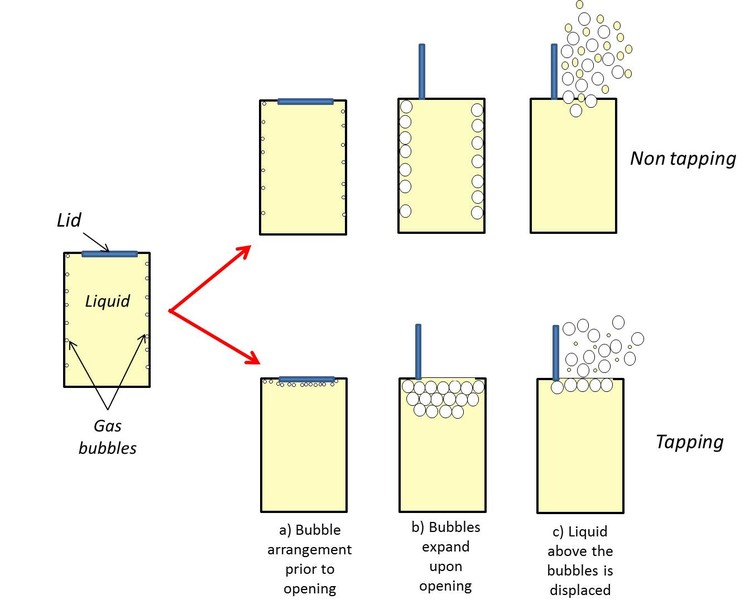
The researcher, Chris Hamlett, a lecturer in Chemistry at Nottingham Trent University, hypothesized that tapping the can before opening it releases some of the bubbles that have settled toward the bottom of the can. As illustrated below, moving the bubbles toward the top means there’s less liquid to pass through before exiting the can, therefore less spillage.
While this all makes sense in theory, chances are it’s not actually true. While Hamlett’s not yet put his tests into motion, representatives from both Coca-Cola and Pepsi have said they aren’t aware of any known results of tapping the can before opening. Likewise, a Snopes.com experiment didn’t find any measurable benefits of tapping.
Now, if you are looking for a proven way to reduce fizz upon popping open a can, stick it in the fridge. A cold can of soda fizzes less than a warm one. Carbon dioxide is more likely to stay “soluble” or dissolved at a cold temperature. The cooler the soda, the less CO2 you’ll have looking to escape and therefore, less chance your soda will spill over the top.
So, there you have it. All these years, it looks like we’ve been tapping our cans for naught.